People have relied on organic salts like tetramethylammonium acetate (TMAA) for decades, not because of a sudden scientific breakthrough, but because researchers kept bumping into limitations with old-school inorganic salts. In the early days of analytical chemistry, labs crowded with sodium or potassium acetates saw their limits pretty fast. When analytical chemists needed better solubility, lower ionic interference, or wanted cleaner, more controlled reactions in aqueous or organic mixtures, tetramethylammonium compounds became more than an afterthought—they became tools. Looking back, I see the story of TMAA following the shift in modern chemistry’s focus toward more precise, tunable reagents. By the 1970s, with growing infrastructure for synthesizing quaternary ammonium compounds, the acetate version found its groove in chromatography and organic synthesis labs. Over time, academic and industrial teams started producing, studying, and tweaking TMAA’s properties, making it a mainstay for sensitive procedures and clever reaction pathways.
Tetramethylammonium acetate stands as a small molecular weight organic salt, usually sold as a crystalline solid or sometimes as a concentrated solution for demanding lab work. Standard labels show its status as a quaternary ammonium salt with an acetate anion, making it energetically stable for shelf storage and robust in handling, but still versatile for researchers. Commercially, people recognize it under various names like TMAA or more rarely “N,N,N-Trimethyl-1-ammonium acetate.” Anyone who’s handled both this and its halide cousins can tell the difference in odor, handling requirements, and solubility—a practical sign that every label conveys.
This salt comes off as white, crystalline, hygroscopic, and easily dissolvable in water and polar organic solvents—qualities that draw attention from those looking to dissolve, extract, or prepare tricky mixtures. It packs a molar mass of around 165 grams per mole, and melts above 200°C, usually with decomposition. Under a microscope, the structure holds up as an ionic lattice tucked between four methyl-capped nitrogen atoms and small but lively acetate ions. Neither oxidizing nor reducing under normal lab conditions, it stays inert until called upon by a talented chemist. No strong odor, low vapor pressure, and a predictable pH in aqueous solution bolster its popularity for high-precision environments. The acetate group can gently buffer solutions, adding a practical angle in bioanalytical and synthetic applications.
Suppliers publish TMAA with tight purity standards, often topping 98-99% assay, measured by NMR or HPLC to keep impurity headaches away. Moisture content gets flagged, as excess water can change stoichiometry in sensitive analyses. Labels highlight storage temperature—cool, dry, and light-protected spaces beat humid benches every time. Standard SDS sheets focus on health and environmental risks, while batch numbers and lot traceability now come standard, reflecting higher industry scrutiny. Chemists tally information about pH value in solution, exact molecular formula, and crystal structure—the kinds of specs that help avoid failed reactions and missteps in regulated settings.
Manufacturers craft TMAA by neutralizing tetramethylammonium hydroxide with glacial acetic acid—two straightforward chemicals, once you get past the caustic nature of the hydroxide. Careful titration yields water and the desired salt, followed by slow evaporation and crystallization. Many producers repeat the purification process, recrystallizing from solvents like ethanol or acetone to get rid of stubborn byproducts and unreacted starting materials. This method underlines the need for precise process control; a slip in ratios or temperatures sidetracks purity and yield. In the few plants I’ve seen, close attention to pH and solvent purity makes all the difference.
TMAA holds up as a mild base and weak nucleophile, thanks to the fully substituted nitrogen atom. This allows the salt to act as a phase transfer reagent, bringing reagents together when other salts just bog down reactions. It won’t jump into redox reactions or readily break down under mild heating, another reason labs keep it handy as a buffer or neutralizing agent. Chemists can swap out the acetate for other anions, passing through anion exchange resins, opening new doors for ionic liquid design. Modifiers use TMAA’s predictable behavior to drive nucleophilic substitutions or act as a counter-ion source for sensitive analytical separations. In a complex mixture, it neither hogs the stage nor disrupts the whole show, instead gently steering reactions and maintaining friendly conditions for lab assays and protocols.
The scientific catalog may call it tetramethylammonium acetate, but trade packaging mixes things up; TMAA, TMAAc, and “quaternary ammonium acetate” all refer to the same chemical. Some catalogs list it as the salt of N,N,N-trimethylmethanaminium and acetic acid, just to keep things formal for regulatory filings. In practice, most research labs know it by its four-methyl acronym, and seasoned chemists learn to scan all synonyms before placing orders to avoid confusion. This patchwork of names grew out of regional branding and changes in naming conventions by various chemical societies and regulatory bodies over the last fifty years.
Handling TMAA does not spark major health alarms compared to many reagents, yet proper caution always trumps bravado. Contact with the skin or eyes brings minor irritation, and an open bottle left in humid air can clump fast, slowing down workflow and contaminating stock. Solid lab practice dictates gloves, goggles, and clean scoops—treating spills with plain water and collecting powder, not creating clouds or flushing down the drain. Most labs I’ve trained in keep TMAA secured with other nitrogenous bases, and clear up spills with a mix of physical clean-up and waste collection, avoiding reactions with incompatible acids or oxidizers. Fire risk remains low, but nobody benefits from careless storage near open flames or strong oxidizers, given any organic compound’s potential under the wrong conditions.
People use TMAA in capillary electrophoresis buffers, ion-pairing agents in HPLC, and as a supporting electrolyte in electrochemical experiments. The chemical steps into molecular biology protocols, sometimes as a better buffer for RNA manipulation than classical sodium salts, which can introduce cation effects. Organic chemists appreciate its ability to solubilize organometallic complexes or activate nucleophilic substitutions. Environmental science labs have picked up TMAA for mass spec analysis of contaminants where metal cations would interfere or mask signals. TMAA does double duty in materials research, acting as a precursor for ionic liquids, where fine-tuning physical properties means the difference between a good and a great product lead.
R&D teams worldwide chase better ways to utilize TMAA, aiming to lower background noise and raise detection limits in modern analytical platforms. Molecular biologists have mined its commercial value by reducing RNase activity in hybridization buffers, a trick I saw speed up a tricky Northern blot. Electrochemists lean on TMAA’s high ionic conductivity and chemical stability for designing better batteries and supercapacitors. Companies tweak the synthetic method by switching to greener solvents or using less energy in crystallization, addressing sustainability and cost pressure at scale. Patent filings show substantial growth in innovative uses over the last decade, reflecting a global effort to squeeze more utility from a simple salt.
Toxicology groups observed that acute oral and dermal exposures to TMAA stay low on severity scales. Chronic exposure studies in rodents show mild, reversible effects at doses above those present in a standard lab. TMAA breaks down to less toxic fragments in the environment, but the quaternary ammonium group warrants attention; metabolites sometimes introduce higher toxicity than the parent compound, justifying strict waste segregation and regulatory reporting. I’ve heard from analytical labs that aquatic toxicity remains relevant, especially downstream of pharmaceutical synthesis plants, prompting stricter monitoring and control of any effluent. Data still accumulates around bioaccumulation in sensitive organisms, pushing regulators to review exposure limits as new studies land.
The path ahead for TMAA will likely ride the trends in green chemistry, better materials science, and precision medicine. Engineers are designing more tailored quaternary ammonium salts for next-gen energy storage; TMAA’s reputation for stability gives it a head start in those prototypes. Analytical chemists keep probing its role in RNA sequencing and epigenetic workflows where contamination or background ions threaten results. The push for sustainable chemicals means more focus on synthetic routes that minimize hazardous byproducts and improve atom efficiency. Pharmaceutical companies investigate TMAA as a part of ionic liquid formulations, searching for bioavailable, less toxic alternatives for drug delivery. As with many unsung lab workhorses, the value of TMAA shows in the steady stream of research papers and patents, never in the hype but in the progress it helps unlock behind the scenes.
Many people outside science circles have never heard of tetramethylammonium acetate. It’s easy to gloss over chemical names, but this substance turns up in corners of research that touch the lives of countless people, whether by pushing forward medicines, sharpening environmental tests, or making industrial processes safer.
Walk into any well-stocked research lab, and you'll find bottles labeled with intimidating long names. Tetramethylammonium acetate is one of those, most often a white powder or clear solution. People in chemistry reach for it when separating mixtures during a technique called liquid chromatography. This precise separation works best with buffer agents that don’t muddy the results. Tetramethylammonium acetate solves this problem through its reliable behavior in water and organic solvents.
Instead of complicating delicate tests, it helps chemists pull out signals that might get lost in noise. That’s important for anyone developing pharmaceuticals or checking environmental contamination, as one wrong note could mean the difference between safety and hidden risks. In my days at a university lab, grabbing the right buffer made sorting out chemical puzzles much less of a headache. Tetramethylammonium acetate earned its spot for that reason.
Modern research places enormous trust in DNA analysis. Whether working on gene therapies, diagnosing infections, or tracking crop diseases, technicians need to cut, copy, and study DNA. Many of these processes depend on small ions that don’t chew up the delicate DNA strands but still let the reactions run their course. Tetramethylammonium acetate brings the positive charges that stabilize the double helix without introducing confusion.
This matters in polymerase chain reaction (PCR), restriction enzyme mapping, and DNA sequencing. The chemical provides just enough structure so fragments sort themselves out, making it easier for scientists to analyze genetic material. As the world looks for rapid testing and personalized medicine, improving DNA research tools isn’t just a lab geek’s concern—it can move the dial on healthcare.
Not all solvents are friendly to the planet. Regulations tighten each year on substances that linger in waterways or produce hazardous byproducts. Tetramethylammonium acetate shows up in greener processes, doing the hard work of dissolving or transferring materials without making the waste problem worse.
Thanks to its solubility in water and its relatively low toxicity, industries use it to replace more dangerous compounds in organic reactions. While not perfect, it is an improvement over some traditional salts that stick around and cause trouble for wildlife. It also helps control reaction rates, giving better yields using fewer hazardous steps. This lines up with the growing push for sustainability in manufacturing and research.
No chemical is completely safe if handled carelessly. Tetramethylammonium compounds can become toxic at high doses, especially for those who work with them every day. Good safety training, modern ventilation, and following safety data sheets protect lab workers and prevent accidental environmental release.
If manufacturers keep aiming for lower waste and more efficient chemistry, substances like tetramethylammonium acetate play a real part in that story. Balancing utility with safety and environmental compatibility sets the tone for progress. In my experience, small changes in chemical choice ripple out and shape the way innovations make their way from lab benches to real-world impact.
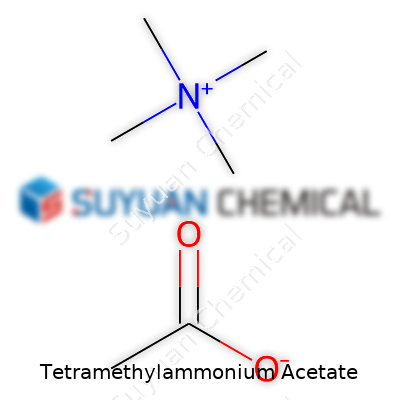
Tetramethylammonium acetate might sound like a mouthful, but it fits into some everyday lab work for a reason. Its formula, C6H15NO2, comes from two pieces: the tetramethylammonium part, which is (CH3)4N+, and the acetate ion, CH3COO–. Put them together, and you’ve got a stable salt known by chemists for its versatility.
The tetramethylammonium group features a nitrogen atom at its core. Surrounding it, four methyl groups (–CH3) attach to the nitrogen, creating a bulky, positively charged ion. On the other side, the acetate ion holds a negative charge, created by a carbonyl (C=O) and a methyl group bonded to a carboxylate (COO–). They balance each other out in the salt, sticking together through ionic interaction. In solution, they separate, but in solid form, these ions arrange themselves to optimize their charges and packing.
During an internship in an organic chemistry lab, I saw firsthand how critical tetramethylammonium acetate can be. We reached for it to adjust solvents, to help drive reactions without tossing in extra water, and even as a mild base. It's less harsh than some other alternatives, making it ideal when you want gentle conditions. The acetate part brings enough flexibility for different synthesis paths. Unlike bulkier or trickier bases, tetramethylammonium acetate dissolves quite well and doesn’t clog up glassware or muddy up reactions. Once, during a tricky methylation, we swapped out another ammonium salt for this one, and it cut down side reactions noticeably.
Researchers often pick this compound for use in phase-transfer catalysis and chromatography. Its strong solubility in water and organic solvents helps it serve as a bridge between different chemical environments, often without the toxicity that some other salts carry. For those doing analytical chemistry, clean reactions and reliable solubility can mean getting accurate results or wasting another day running repeats. In bioorganic synthesis, the low reactivity of the tetramethylammonium ion keeps things gentle enough not to ruin delicate biomolecules. If you’ve slogged through a reaction that fell apart simply due to choice of base, you’ll know the relief a predictable salt like this provides.
No chemical is perfect. Handling tetramethylammonium acetate takes a bit of attention: Some sources mention mild toxicity, so disposals need to follow best practices. Spills, if ignored, can leave sticky residues hard to clean off benches. Keeping it sealed helps, especially to avoid moisture which can make it clump. Labs aiming to size up green chemistry efforts could look at further reducing waste and substituting solvents, but tetramethylammonium acetate often stacks up well compared to harsher alternatives in terms of safety and environmental profile. Revising protocols once a year helped us minimize unnecessary use and improved storage in our lab, reducing both exposure and waste.
Tetramethylammonium acetate remains a dependable tool, valued for performance in sensitive conditions, and its straightforward chemical nature means chemists can rely on it during both routine and specialized procedures. Beyond its chemical formula, experience in the lab shows its value is more than theoretical—it’s practical, making life a bit easier for those doing the work.
Tetramethylammonium acetate turns up often in my lab notes, tucked among solvents and buffers. This reagent deserves respect: beyond its morning-bagel-sounding name hides a material that can stir up challenges in any chemical storeroom. People sometimes forget: it looks plain, but its risks carry weight.
Opening a fresh drum of tetramethylammonium acetate can stir up both dust and worry. The powdery or crystalline solid kicks up much more easily than bigger, clumpier chemicals. I never forget my goggles and gloves here. Contact with skin or eyes stings, but the risk ramps up if someone breathes it in or lets it linger on hands. Researchers have reported irritation, coughing, and nasty headaches—in extreme cases, poisoning can threaten.
So, I reach for my proper gloves—nitrile, not those useless thin latex ones that snag and let through the smell of chemicals. Eyes don tight protection. I keep face shields handy, especially while decanting bigger amounts, since a curious gust of air can send crystals flying. Pouring turns messy in a busy environment, so it makes sense to keep the working area quiet, with the chemical under a fume hood.
Let’s talk storage. This isn’t salt for the kitchen shelf. Fast temperature swings and bright light start breaking down the chemical—or at least turn the label-writing on its side. I store tetramethylammonium acetate in a cool, dry cabinet, away from sunlight, acids, or oxidizers. Humidity is the enemy here: even modest moisture in air turns this powder into a sticky, half-dissolved mess.
Air-tight containers—think well-fitted plastic or glass with solid seals—hold up against air and water. I avoid crowding the shelf, since a toppled bottle means spills and clean-up, not to mention a waste of material and extra risk. Every container wears its own clear label: name, date, concentration or batch, hazard pictograms.
Most of us learn from near-misses. I remember cleaning up a small spill that left my gloves feeling oddly warm. Turns out, this chemical reacts with strong alkalis or oxidizers, and a bit of bleach from a nearby cleaning rag kicked up an instant fizz. I wiped it up fast, switched out gloves, and reported the incident. If someone breathing the dust or liquid feels cough or sore throat, I send them for clean, cool air and get them medical attention as fast as possible.
Emergency showers and eyewash stations matter. Nobody expects to use them, but a splashing accident sorts the careful from the careless. My rule: before even opening the container, I check that the eyewash works and that spills will flow toward drains, not toward coworkers.
Having procedures on paper means nothing unless people know them by heart. I help train each newcomer by walking them through real practice—handling, storage, what to do if a spill surprises them. The team checks up on each other. No lone-wolf behavior in dodgy corners of the lab. Detailed safety data sheets hang on walls and digital storage.
Staying safe with reagents like tetramethylammonium acetate involves more than careful reading. It relies on steady habits—using the right gear, storing the chemical right, staying ready for trouble, and making sure every team member feels responsible for each other’s safety every day.
Tetramethylammonium acetate often turns up in labs, especially during special syntheses or as a reagent in organic chemistry. Its formula looks harmless enough — a simple acetate tied to a quaternary ammonium group. If you’ve worked in a chemistry lab, you know every compound brings its own baggage. This one, too, calls for a fair bit of respect.
Handling chemicals without getting the full picture is where I’ve seen most problems start. Many users assume that “ammonium” or “acetate” in the name means it’s mostly benign, lumping it together with kitchen vinegar or ammonium chloride. That’s a trap. Tetramethylammonium compounds, especially when they break apart, can trigger dangerous effects in the body. Ingesting or getting exposed to enough of it can hurt the nervous system and the heart, because the tetramethylammonium ion interferes with the natural signals nerves send. There are reports of people collapsing, convulsing, or worse, after coming in contact with its close relatives.
Some folks want to sweep toxicity under the rug since acute deaths reported are rare compared to other lab chemicals. That doesn’t mean it gets a free pass. Its toxicity means anyone working with it ought to be on guard, even if only small volumes are involved. Common sense says gloves and a hood aren’t up for negotiation. Even skin contact can pose problems, as the ion can slip through and start causing symptoms.
Most chemists watch for spills, but the bigger issue is where waste ends up. Tetramethylammonium compounds don’t just break down in the drain. If they get into waterways, they can cause problems for aquatic life. Fish and invertebrates have systems vulnerable to these ionic disruptors, so those routine drain tossings add up over time. I’ve seen plenty of labs improve by storing and then sending their waste out with professionals, rather than hoping water treatment plants can handle it without a hitch.
Some stories still stick with me — one summer a graduate student missed the warning on a reagent bottle, ended up in the ER after a spill. That case pushed our department to run regular safety refreshers and double-check all our labeling. The lesson wasn’t buried on a memo: everyone started informing newcomers right at the bench, not just in safety videos.
Proper ventilation, solid gloves, and face shields aren’t fancy extras; they’re part of daily work. Waste disposal means a call to licensed collection, not a shortcut down the drain. It helps to keep a well-stocked first aid station close by, with people who know what to do in case exposure happens.
That call for diligence doesn’t mean Tetramethylammonium acetate belongs on a banned list, just at the center of careful lab work. Rushing or slacking in procedures stacks the risks. By knowing the hazards and building habits for safety, labs can keep turning out good science without turning researchers into patients.
Tetramethylammonium acetate is a chemical folks in labs see a lot, whether they're developing new batteries or tweaking reaction conditions for better yields. One thing draws attention over and over: its willingness to dissolve in water. Load it into a beaker with some distilled H2O and you’ll see it blend in right away—no stubborn clumps at the bottom, no need for violent stirring. The acetate part helps keep things friendly with water molecules, lots of ionic character, lots of chance for hydrogen bonding to kick in.
That ease of dissolving isn’t just for show. Scientists count on it when preparing buffers for biological research, or when plotting out synthesis runs where even little bits of undissolved salt could throw off the whole game. Turns out it can also handle itself in some alcohols, like methanol and ethanol—a handy bonus for certain organic reactions where using water would spoil the outcome.
Back in college, I worked with tetramethylammonium acetate in a project on peptide mapping. Every day started with dissolving measured amounts of salt into buffer, always watching for cloudiness or leftovers. Not once did it let us down. That reliable dissolving power matches up with published reports: scientists have found that you can get gram after gram into water without trouble. This contrasts with other ammonium salts, where you might face restrictions based on both polarity and the size of your organic tag.
Chemists take advantage of its solubility for chromatography, mass spectrometry prep, and making sure proteins don’t clump together during experiments. In some electrochemical uses, quick dispersion in solvent speeds up the setup process and improves consistency from one test to the next. With the right solvent and temperature, tetramethylammonium acetate proves flexible, mixing into aqueous or some non-aqueous systems, though it stalls out in truly non-polar solvents like hexane.
Solubility affects more than a reaction flask. In labs with limited waste controls, water-soluble chemicals can slip into wastewater streams. Managing that risk means putting real waste treatment steps in place. You can’t overlook it, especially as environmental rules tighten. Reliable testing, good data records, and investing in routine training protect both people and the groundwater outside.
The chemical carries modest toxicity—breathing in fine dust, or letting it soak into the skin for too long, isn’t wise. Gloves, good ventilation, sharp eyes on the safety sheet: that’s basic lab smarts.
More open-access research on solubility across temperatures and mixed solvents would help the next crew of scientists plan experiments faster and safer. Chemical suppliers should keep sharing up-to-date info, including real-world safety notes from labs across industries. Investing a little time up front in understanding how tetramethylammonium acetate dissolves pays off with fewer headaches and better results all around.
Spotting the right solvent now means fewer failed runs later and less wasted effort troubleshooting mystery lumps at the bottom of a reaction tube. That saves money and keeps operations running smoother.