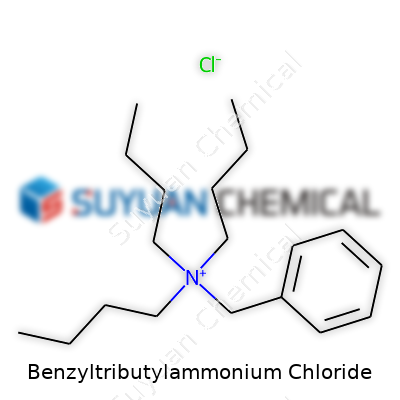
Curiosity about quaternary ammonium salts ramped up in the twentieth century, especially as scientists started searching for practical phase-transfer catalysts. This led to the synthesis of Benzyltributylammonium Chloride (BTBAC), a salt where a benzyl group joins three butyl chains around a nitrogen atom. Companies soon realized its value, not just for its chemistry but for what it did for more efficient industrial processing. While early work focused on basic organic syntheses, BTBAC soon found a place in scale-up pipelines. The rise of more complex organic reactions made the need for strong yet manageable phase-transfer agents like BTBAC more acute. The compound’s story isn’t about big flashes or isolated discoveries; it’s more about the steady push for better tools that turn stubborn lab reactions into scalable reality.
Benzyltributylammonium Chloride isn’t flashy at a glance—it’s a colorless to pale yellow liquid or solid, depending on temperature and humidity. Its structure, a robust benzyl group tethered to a quaternary ammonium head, gives it a beachhead at oil-water interfaces, which means it can ferry ions across boundaries that usually keep them apart. BTBAC stands out for its remarkable ability to bring together water-loving and oil-loving chemicals. Most supply houses deliver it as a solution, typically about 50% concentration in water or an organic solvent. The compound’s odor lingers—dim but unmistakable—and its viscosity feels just right for industrial manipulation. Reliability and consistency of melting behavior matter here more than in most bottles on the shelf.
Benzyltributylammonium Chloride presents as a slightly viscous, pale liquid under room conditions. Its melting point sits just below room temperature, which makes it pourable, and it shows good solubility in water, alcohol, and a host of organic solvents. Chemical stability ranks high; it resists breakdown under normal handling, though strong acids or bases eventually degrade it. The molecule carries a positive charge on its nitrogen, balanced by a single chloride anion—a pairing that dictates how it moves between phases in reactions. Most labs report a molecular weight near 340.0 g/mol. Thermal decomposition doesn’t happen until higher temperatures, so normal industrial conditions rarely trouble it. This resilience, plus its strong ionic character, explains much of its popularity in both bench and plant work.
Technical grade BTBAC generally comes with purity above 98%. Impurities can include bits of unreacted benzyl chloride or tributylamine, both easily distinguished on standard quality checks like TLC or HPLC. Product bottles bear hazard codes due to its irritant properties, with labeling demanding gloves, goggles, and ventilation in the workspace. Labels show crucial batch numbers, expiry, and recommended storage conditions—typically below 25°C, away from light and moisture. Transport containers need secure caps, sometimes with secondary containment in case of spills. Most suppliers print solvent content and pH right on the outside for quick assessment and easy reference on loading docks and in chemical cages.
Production of BTBAC usually involves reacting tributylamine with benzyl chloride in a simple alkylation reaction. The process runs best using an aprotic solvent like toluene or acetonitrile, helping the two starting materials meet. Stirring and gentle warming encourage the benzyl chloride to snap onto the amine. Quenching happens with chilled water, leading to immediate precipitation or separate phases. Final product washes help remove leftover reactants, and a drying step clears out water and stray volatiles. Scale-up in an industrial setting needs continuous monitoring for vapor leaks, since tributylamine and benzyl chloride both release fumes that can irritate mucous membranes.
BTBAC shines as a phase-transfer catalyst—moving anions from water into organic solvents, where otherwise sluggish reactions pick up speed. Take nucleophilic substitutions or certain alkylations: BTBAC lets them run under milder conditions, saving money on energy and raw materials. Chemists have pushed the boundaries by tweaking its side chains, swapping out the benzyl or butyl groups to tune solubility or reactivity. Even the chloride can swap for other anions, leading to a tailored catalyst for specific needs. Its action looks simple, but it transforms entire workflows by linking water-phase reagents with organic chemicals that otherwise refuse to talk to each other.
BTBAC answers to several aliases: Benzyltributylammonium Chloride, Benzyltri-n-butylammonium Chloride, and BnNBu3Cl in shorthand. Some suppliers refer to it as Triton B or TBBA. Trade names rarely shift the core product but may indicate formulation differences, especially between lab-grade and sterile or pharmaceutical batches.
Toxicity studies put Benzyltributylammonium Chloride into the moderate risk category. It irritates eyes, skin, and lungs, meaning researchers treat it with respect—gloves, goggles, and fume hoods form the frontline. Spills call for careful blotting and quick dilution with water before collection for hazardous waste. Fire risks stay low under routine handling, but since it decomposes with heat, closed vessels and careful temperature control matter. Long-term storage away from acids prevents unwanted degradation. Workers train on emergency procedures before ever handling BTBAC—I’ve watched safety trainees practice spill drills using less toxic simulants before touching real stock. Standard operating procedures stress step-by-step checks for tight seals, clean tools, and posted emergency contacts, building muscle memory for safe routine handling.
Industrial manufacturing and research labs both keep Benzyltributylammonium Chloride on hand for phase-transfer catalysis. In pharmaceutical synthesis, BTBAC unlocks reactions that would otherwise crawl to a halt in water-only or organic-only settings. The agriculture sector relies on it to develop pesticide intermediates, and water treatment plants use it for ion exchange processes due to strong affinity for specific contaminants. Dye manufacturing benefits from its action in the coupling of aromatic amines. Electroplating solutions sometimes include BTBAC for smooth coatings. From my time spent in various process labs, the best outcomes always came by taking full advantage of its phase-hopping skills, letting reactions finish faster and with less waste.
Each decade, BTBAC’s role broadens. Climate-driven interest in green chemistry has research groups hunting for catalysts that work at lower temperatures and in water-based rather than organic solvents. BTBAC fits that bill. Scientists now design entire reaction platforms around phase-transfer chemistry, building new drugs and materials in ways that use less energy and create fewer byproducts. Undergraduate chemistry courses regularly train future researchers on BTBAC-based reactions, knowing this experience translates directly into competitive skillsets. Ongoing R&D looks at not just new reactions, but also ways to recover and recycle BTBAC for sustainability. In my own lab work, I’ve seen pilot programs cutting catalyst waste by half with closed-loop systems. This approach points the way toward practical, scalable, and safer chemical manufacturing pipelines.
BTBAC’s toxicity earns it respect and careful handling. Animal studies reveal irritation at moderate concentrations and show higher doses can cause organ stress over time. Because its structure resembles other quaternary ammonium compounds, environmental chemists track its fate in wastewater streams. Regular workplace monitoring using air and surface sampling helps keep exposures well below regulatory limits. Personal experience with safety audits reinforced my respect for company protocols—no one wants a chronic exposure scenario. New studies keep expanding our understanding of its long-term impacts, both for workers and the environment, which drives the push for safer substitutes or improved waste treatment.
Regulatory trends and sustainability goals influence BTBAC’s industry trajectory. Equipment upgrades and stricter guidelines keep exposures down, and improved formulations lower the need for fresh chemicals with each batch. Green chemistry continues to nudge process engineers away from persistent or high-toxicity phase-transfer agents, but BTBAC won’t vanish soon: its versatility still solves plenty of current challenges. Research into biodegradable analogs advances every year, yet cost and performance must catch up before wholesale changes roll out. Automation and in-line monitoring offer tighter control over BTBAC use, recycling, and waste management. As new reaction families and industrial applications emerge, BTBAC keeps its relevance by delivering practical, familiar, and well-understood chemistry.
Benzyltributylammonium chloride rarely turns up in conversations outside chemical labs, yet its impact stretches far past the textbooks. Anyone who’s handled water treatment processes or followed how medications reach their final, usable forms has quietly relied on chemicals like this one. Labs and manufacturing floors depend on it as a phase-transfer catalyst—a fancy term for something that helps oil-soluble and water-soluble substances mingle long enough for important reactions to happen. In practice, that means more efficient manufacturing and less energy wasted, drawing real value for producers and, by extension, consumers of all sorts of household products.
Many chemists I know trust benzyltributylammonium chloride when faced with reactions that would otherwise be nearly impossible on a practical scale. Picture a pharmaceutical company trying to make a heart medication that starts with a water-based ingredient and an oil-based one. Normally, they’d struggle to get these materials talking to each other. This chemical steps in as a translator—one that can smooth out wrinkles for chemical syntheses involving things like nucleophilic substitutions or oxidation reactions. I’ve seen research teams clear major bottlenecks thanks to this simple addition to their protocol.
My own city relies on advanced water treatment, and specialists in these plants point out that removing pollutants doesn’t just involve filters and tanks. Chemicals like benzyltributylammonium chloride find their place in purifying drinking water and managing industrial wastewater, primarily in the breakdown of stubborn organic contaminants. Not every treatment process needs such additives, but the ones that do draw real benefits in terms of reach and reliability. In my experience speaking to environmental chemists, efficiency in pollutant breakdown means safer water at the tap and more sensible long-term costs.
For all its advantages, working with specialty chemicals always brings responsibility. Benzyltributylammonium chloride, like many catalysts, comes with risks if workers don’t follow correct handling and disposal protocols. Stories from chemical plant staff and academic researchers alike underline how exposure without adequate protective gear can cause skin, eye, or respiratory irritation. Companies willing to invest in regular staff training, proper PPE, and transparent reporting processes tend to avoid costly accidents and build trust within their teams and with the communities they serve.
Several research teams across the globe focus on green chemistry—reducing unnecessary chemical waste and finding safer alternatives without sacrificing performance. These efforts connect directly to the use of phase-transfer catalysts. By choosing approaches that minimize hazardous byproducts and cut down on energy use, chemical companies are stepping up to embrace smarter production. Solutions include ongoing education for lab technicians, investments in improved ventilation systems, and collaboration with environmental scientists to design cleaner processes from the outset. Companies that focus on these details not only protect their local ecosystems but also gain a business edge as sustainability moves from buzzword to requirement.
None of this matters if we don’t stay curious about what shapes our world—right down to obscure chemicals like benzyltributylammonium chloride. Staying informed and open to innovation keeps manufacturing safer, everyday goods more affordable, and environmental impact under control. That’s the kind of chemistry that makes a difference, even outside the lab.
Benzyltributylammonium chloride carries the chemical formula C19H34ClN. It comes together from a benzyl group, three butyl chains, a central nitrogen atom, and a chloride ion for balance. The entire structure holds up strong ionic character, giving it a unique position in organic chemistry labs and industrial applications. Seeing that formula on a label means you’re working with a quaternary ammonium salt packed with both organic and ionic qualities—a true workhorse for those in synthesis and extraction work.
I still remember crunching through a tangle of organic syntheses in college. The first time I saw benzyltributylammonium chloride in a protocol, it handled the transfer of ions between water and oil like no other. This wasn’t just another chemical—it allowed reactions to break through the boundaries of phase separation, moving chlorides or nitrates right where they were needed. This is what chemists call a phase-transfer catalyst. Without it, hours in the lab might stretch into wasted days trying to coax polar and non-polar reactants to cooperate.
The presence of both organic and ionic parts means C19H34ClN dissolves in a surprising range of solvents. Paint manufacturers, pharma research teams, and analytical chemists reach for this compound because it helps to shuttle ions back and forth across otherwise incompatible phases. That means improved yields, cleaner reactions, and more efficient extractions. I’ve seen how just a tad of benzyltributylammonium chloride pulls stubborn ions from an aqueous solution into an organic phase, making purification steps smoother and reducing chemical waste. Every researcher who’s had to separate a desired product from a contaminated mess can confirm the value that brings.
Every powerful tool comes with a need for caution. Benzyltributylammonium chloride’s chemical formula points to a structure that isn’t exactly mild in the lab. That long carbon chain—typical of many quaternary ammonium compounds—makes it hazardous if misused. It acts as a skin and eye irritant, so gloves and goggles aren’t optional. Proper storage matters as well, given that it breaks down over time if it picks up moisture or heat. If chemical hygiene slips, you get exposure risks and unwanted byproducts, which put a stop to even the most promising reaction scheme.
To keep labs and production lines safe, regular training on storing and handling benzyltributylammonium chloride pays off. Monitoring for airborne particles, using closed transfer systems, and updating safety data sheets whenever new findings emerge helps avoid accidents. Downstream, waste disposal presents challenges. This chemical can persist in water and soil, causing concern for environmental agencies. Investing in greener phase-transfer agents and supporting studies on biodegradable alternatives carves a responsible path forward. Active steps on both safety and waste frontlines keep scientific progress on course without leaving a toxic footprint.
Chemists and manufacturers keep reaching for benzyltributylammonium chloride’s unique formula, thanks to its blend of solubility and catalytic muscle. As demand for efficient and cleaner chemistry grows, new approaches to minimize hazards and improve breakdown after use will shape its future. C19H34ClN has more to offer—but that potential depends on respect for its power and a commitment to continuous safety improvements.
Benzyltributylammonium chloride comes up in a lot of industrial chemistry conversations, mostly because it helps move molecules around in reactions that wouldn’t behave otherwise. People use it as a “phase transfer catalyst,” which just means it helps ingredients mix that don’t like to get along. You’ll see it in labs, chemical plants, and sometimes in the process of making medicines or plastics.
Safety information matters whenever someone works with chemicals, and this one grabs extra attention. Benzyltributylammonium chloride can cause skin and eye irritation. Breathing in its dust or fumes may irritate your nose, throat, and lungs. Swallowing it can bring nausea, burning sensations, or worse. Most of those using it at work learn to respect these outcomes pretty quickly.
The European Chemicals Agency put its cards on the table, calling it an irritant, both to skin and eyes, with the potential for “serious eye damage.” The U.S. National Library of Medicine flags it for the same issues. Not exactly a friendly substance to have around at home or anywhere kids get curious.
Years ago, I watched new lab workers handle this substance with their bare hands. It didn’t take long before someone regretted skipping gloves. Benzyltributylammonium chloride sticks to skin. It doesn’t just wash away. A burning feeling can linger, a reminder not to let it touch you in the first place. If you get it in your eyes, medical attention becomes the first priority.
Long-term data on cancer or genetic risks isn’t strong, but repeat exposure doesn’t sound smart. Like other quaternary ammonium compounds, accidental inhalation (which happens more than most folks admit) can hit your lungs hard, especially if you already struggle with asthma or allergies.
Chemicals such as this one can slip out during manufacturing, and nobody wants to see toxic runoff in streams. Fish and other water-dwellers don’t handle these compounds well. They can build up inside the food chain, then trickle back up to humans. European regulators track emissions, pushing industries to control leaks and clean up spills quickly. Companies that get sloppy face heavy fines, sometimes even bigger headlines.
The key is training. Gloves, goggles, proper ventilation, and chemical hoods block most problems. Every bottle or drum should spell out hazard symbols in bold print. Spills get neutralized right away. Disposal follows strict rules, with nothing washing down the drain. If something goes wrong, safety showers and eyewash stations must be ready.
Innovation will help over time. Green chemistry approaches aim to replace or reduce the use of hazardous catalysts wherever practical. Some companies already try alternatives with less sting and fewer environmental risks. It doesn’t solve every challenge, but it means the industry is paying attention.
No chemical in the workplace gets a free pass. Benzyltributylammonium chloride serves useful roles and gets handled by people who know the drill: treat it with respect and learn the signs of trouble. Regulations exist for a reason. Solid safety habits make the difference between an ordinary shift and a trip to the ER. Staying informed and cautious doesn’t only protect workers—it also keeps the community and environment a little safer.
Benzyltributylammonium chloride isn’t a substance people stumble across daily, but in labs and industrial settings, this quaternary ammonium compound has become a familiar name. It shows up in organic synthesis, phase transfer catalysis, and sometimes even in environmental labs. One lesson I learned early in my time as a lab tech: storing chemicals the wrong way quickly turns from headache to hazard.
Benzyltributylammonium chloride reacts poorly to excess heat and moisture. Left unchecked, this can leave folks dealing with degraded product or—worse—safety issues. I recall once finding a similar quaternary ammonium salt clumped from sitting near a hot water line. Not only did it lose effectiveness, but it also created unnecessary cleanup and inventory headaches.
This compound should stay in tightly closed containers—preferably the ones it shipped in if possible. Keeping air and water out stops it from absorbing moisture, which in turn helps prevent caking and breakdown. Each time someone opens and shuts a bottle, air sneaks in, so it pays to keep those bottles closed unless measuring out an exact portion. One colleague left a cap loose on a Friday afternoon. By Monday, that bottle was a solid lump—almost impossible to use without a fight.
Find a space free from humidity, such as a dry cabinet or desiccator. A standard chemical storage cabinet that’s cool and away from bright sunlight often does the trick. Direct sunshine or heat from nearby equipment could warm up the room enough to encourage decomposition. Toxic fumes and skin irritation aren’t worries to take lightly. I’ve seen small leaks from overfilled or improperly sealed bottles cause a whole lab to clear out.
Clear labeling matters. I’ve seen storage shelves where the only distinction between two bottles was a faded sticker. A clear, readable label stops anybody from making mistakes in a hurry—especially when rushing through routine tasks. Segregating chemicals by hazard class can make a difference in emergencies. For instance, keeping this chloride away from strong oxidizers and acids makes sense, considering unexpected reactions can occur if spills happen together.
Bins marked for hazardous chemical waste can’t be an afterthought. Even small amounts of leftover benzyltributylammonium chloride should go into approved disposal. Pouring any chemical down the drain never works out for anybody. Many facilities now use logbooks for waste streams, and I’ve seen surprise inspections where proper labeling and secured lids kept us out of trouble. A single carelessly dumped beaker can lead to department-wide consequences.
Trusting that a chemical supplier shipped in a sealed, stable bottle works most days—until it doesn’t. Inspect shipments upon arrival and jot down details in a log. This habit caught a damaged container for us once and likely avoided both product loss and cleanup headaches.
In the end, storing benzyltributylammonium chloride comes down to respect for chemistry and the people handling it. Safe, dry, labeled, watched—that’s the approach that kept my lab running smoothly, costs down, and everyone breathing a bit easier at the end of the day.
Benzyltributylammonium chloride shows up as a white to off-white powder or crystalline solid. Most lab workers spot it by its faint yellow tinge if the batch isn't fresh or absolutely pure. This isn’t stuff that dazzles with shiny crystals or abrupt color—don’t expect a showpiece. Its appearance leaves nothing to guess: touch it and you feel a slight greasiness, a sure sign of its organic salt character.
Folks who’ve handled enough organic salts notice how moisture from the air will soften benzyltributylammonium chloride in humid climates. If you’re working in a small, hot lab without air conditioning, you might see a lumpy mass in your reagent jar before a boilerplate white powder. This makes it clear to store the material with care, away from open air and sealed tight after using it. Storing it near a window or in a damp environment just invites clumping and worsened handling. Students often learn the hard way that a scoop left out overnight doesn’t look the same in the morning.
One thing about benzyltributylammonium chloride: it mixes well with both water and organic solvents such as chloroform. Its solubility in water stacks up at around 20-50 grams per 100 milliliters at room temperature—definitely soluble, but not "instant dissolve" like table salt. What makes it stick out is a willingness to mix with more than just water. You toss it into an organic solvent, and it blends in neatly, which is why so many chemists turn to it in synthesis work. For example, extracting ions or transferring them from one liquid phase to another needs a chemical that sits comfortably in two worlds. This salt fills those shoes.
Watch out for excess humidity because benzytributylammonium chloride draws water from the air. Clumps in your sample? That’s moisture. If you're aiming for pure measurements or clean reactions, dry the compound ahead of time or weigh it immediately after grabbing your portion. Sometimes, storing it with a drying agent avoids this trouble entirely.
Across many labs, this salt turns up in phase-transfer catalysis. Any synthetic chemist will tell you it speeds up some reactions and enables certain transformations that might not run cleanly otherwise. That solubility in both water and organic solvents isn’t just a trivia point in a textbook—it’s the reason the compound actually gets used.
In the field, mishandling stands out as a real, everyday problem. A damp workbench or careless storage means the salt loses its crisp texture, becomes sticky, or forms lumps. This impacts measurements, which in research and production becomes costly fast. Every ruined batch teaches the same lesson: moisture matters. So does cleanliness. Keeping the lid tight and bottles properly labeled in a cool, dry place should be non-negotiable. Occasional checks of your stock don’t hurt either.
Benzyltributylammonium chloride, given its ready solubility, needs careful handling, especially in large quantities. Spills on the bench don't just waste material—they can create slippery surfaces. Labs benefit from posted reminders and training, not just for correct storage, but on cleanup and disposal practices. Protective gloves and eyewear always help, since salts like these can annoy skin given enough contact.
Having worked in small and large labs, I’ve seen rushed lab techs turn the white powder into an afterthought on cluttered shelves. Focused routines, labeled and sealed containers, and a healthy respect for humidity make the difference between smooth days and frustrated ones. Benzyltributylammonium chloride isn’t glamorous, but it performs when treated with thought and care.